Knowledge Management
OK, so what exactly is knowledge management (KM)? It seems like we should cover it here since Q10 has become the hottest topic in our industry at the moment as FDA moves forward with the QMM protocol assessment in response to increasing drug shortages. Q10 is finally getting the attention it deserves, what a pity that it took over a decade…
Just for background, KM is one of the two “enablers” within ICH Q10 that may permit a site to achieve an advanced (or mature) Quality System. This is a site who reliably produces high quality medicines “without extensive regulatory oversight”.
The concept of KM is defined in ICH Q10 as follows:
Product and process knowledge should be managed from development through the commercial life of the product up to and including product discontinuation. For example, development activities using scientific approaches provide knowledge for product and process understanding. Knowledge management is a systematic approach to acquiring, analyzing, storing, and disseminating information related to products, manufacturing processes, and components. Sources of knowledge include, but are not limited to, prior knowledge (public domain or internally documented); pharmaceutical development studies; technology transfer activities; process validation studies over the product lifecycle; manufacturing experience; innovation; continual improvement; and change management activities.
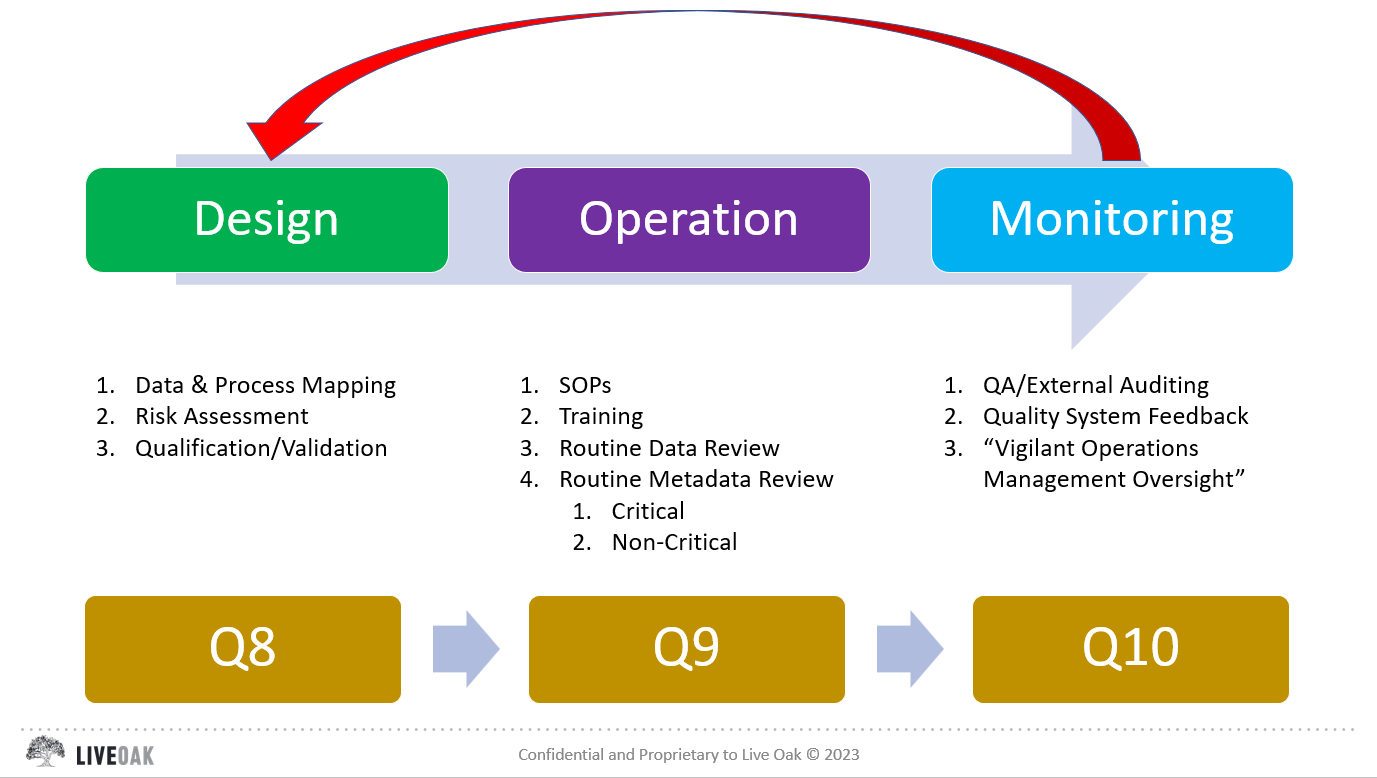
Sounds great, but a definition without practical examples is difficult to envision, especially when operating within a highly regulated environment and very limited resources. I have been focusing on this concept for the past few months, trying to develop a means for explaining KM that is “non-invasive”, so as to not disrupt ongoing operations. I have come to the conclusion that KM can be implemented in various ways, however, in my opinion, the most efficient and effective way is to capture knowledge (explicit and implicit) within our workflow validation packages (see Q2 of FDA’s Data Integrity Guidance). A workflow validation package includes consideration of hardware/software/personnel/documentation, and implements a risk based strategy for ensuring the accuracy and completeness of data via the three pillars of governance: Design – Operation – Monitoring
Example #1: An analyst knowing that an extra minute of sonication during sample preparation reduces variation in assay results is potential knowledge that can be integrated into the quality system to reduce OOS’s and drug shortages. However, this will remain “information” in the analyst’s head until there is a management strategy in place to document this implicit information within the workflow validation risk assessment, at which point it then becomes knowledge. Information does not become knowledge until it is documented, and the most efficient capture of this information is within a workflow validation risk assessment! This forms the library of knowledge that can be used throughout the organization when needed (in this case, during an OOS root cause evaluation). The ability to assign a potential root cause by referencing the KM library will improve efficiency and demonstrate the “scientific rationale” necessary to perform re-testing/sampling.
Example #2: It is explicit information that failure to rinse the probe prior to pH analysis will cause contamination of the sample, lowering the pH and possibly resulting in an OOS. This is just science. This will remain information, however, until there is a management strategy in place to document this implicit information on the workflow validation risk assessment, at which point it then becomes knowledge. This forms the library of knowledge that can be used throughout the organization when needed (in this case, during an OOS root cause evaluation). The ability to assign a potential root cause by referencing the KM library will improve efficiency and demonstrate the “scientific rationale” necessary to perform re-testing/sampling.
Without a KM library to reference, the regulators often conclude the firm is relying on their opinion to justify root cause conclusions/product impact statements/rationale for re-testing/other critical GMP functions. This is the “root cause” of TNTC 483s!
It is time to get on board with KM – it’s a “no-brainer” 🥸
Pete